37 cr3+ orbital diagram
Simplified MO energy level diagram for Cr(CO)6. Note the empty π* orbitals. Figure 3-1 Molecular orbitals of Cr(CO)6 (Only interactions between Ligand (σ- and π*) orbitals and metal d-orbitals are shown.) Diamagnetic Thermally and air stable Reversible Fe2+/Fe3+ potential. Click hereto get an answer to your question The electronic configuration of Cr^3 + is: 1s22s22p63s23p64s13d5 which is half-filled d-orbital. Cr3+ has 3 electrons removed from the outermost shell. Therefore, the electronic configuration comes out to be [Ar]3d3.
A bare molecular orbital diagram is presented and you must drag the correct orbitals and labels onto the diagram. hexaquacalcium(II) - [Ca(H2O)6]2+. hexaquavanadium(III) - [V(H2O)6]3+. hexaquachromium(II) - [Cr(H2O)6]3+.

Cr3+ orbital diagram
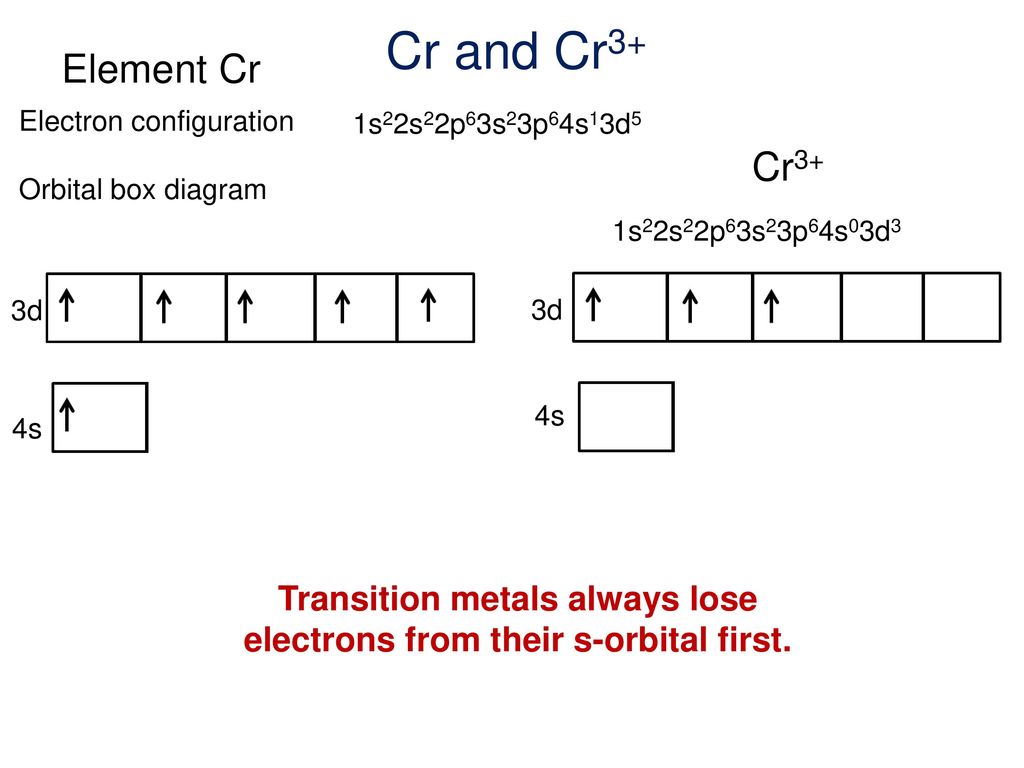
Chromium(III) ion. Molecular Formula Cr. Average mass 51.994 Da. Cr3+. Chromic cation. Chromium ion. Video: Cr, Cr2+, and Cr3+ Electron Configuration Notation. • In writing the electron configuration for Chromium the first two electrons will go in the 1s orbital. Partial Orbital Diagrams and Condensed Configurations. A partial orbital diagram shows only the highest energy sublevels being Write the condensed electron configuration for each atom, recalling the irregularity for Cr. Since there are 3 d electrons they are all unpaired. Cr3+ is paramagnetic.
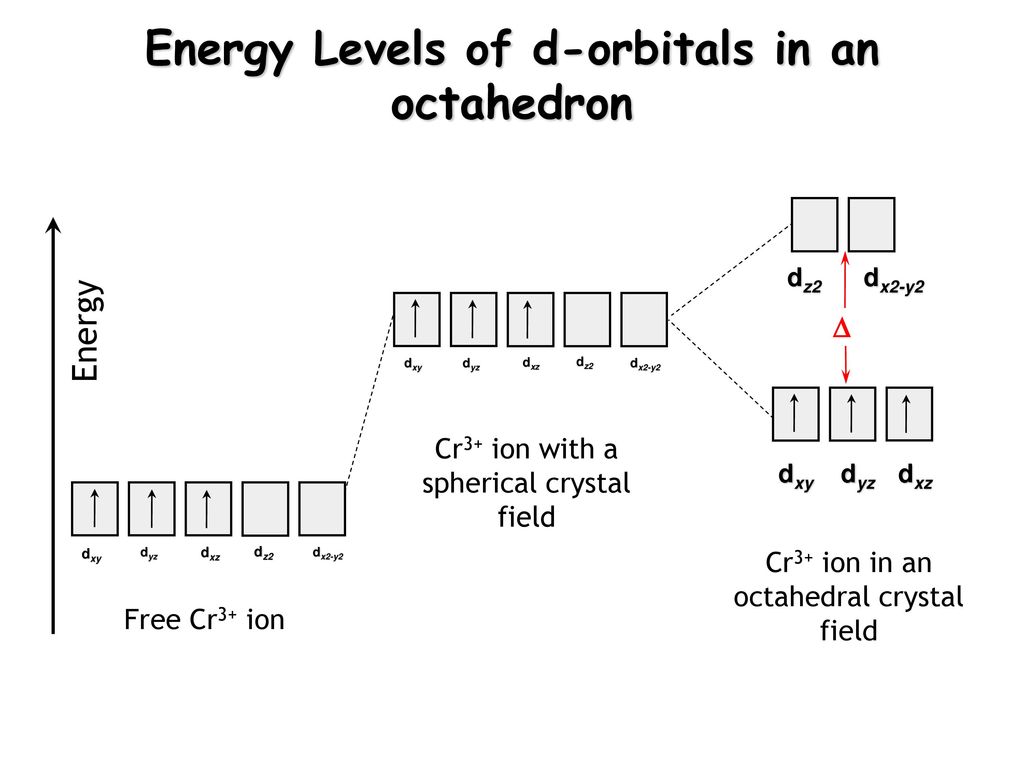
Cr3+ orbital diagram. 3+ [9]. Thermal (heat) conductivity. - - Orbital Diagram for Chromium. Atomic Data of Chromium (Element 24). Valence electrons. It turns into green oxide (Cr2O3) when heated [3]. Write orbital diagrams for each of these ions. A. V^5+ B. Cr^3+ C. Ni^2+ D. Fe^3+ E. Determine if the following ions are diamagnetic or paramagnetic. V5+ orbital diagram keyword after analyzing the system. Cr3+ has 3d5, 4s1 orbital, d10 and d5 are most stable orbitals. This will be explained in crystals field splitting theory. The red line is the Frost diagram for Mn in acidic conditions (pH 0); the blue line is in basic conditions. A few explanations will make the diagram clear The Ti3+, V3+, and Cr3+ complexes have one, two and three d-electrons respectively, which fill the d-orbital energy diagrams for high and low spin Co2+ complexes, d7. Rules of thumb In the case of Cr3+ and V2+, the energy penalty for distorting the complex away from octahedral symmetry - to...
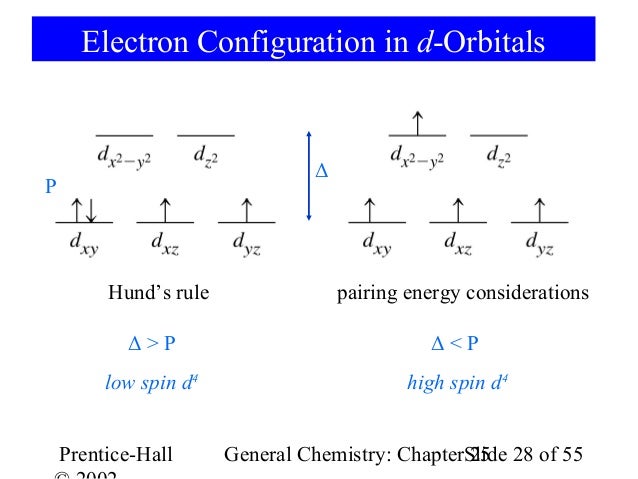
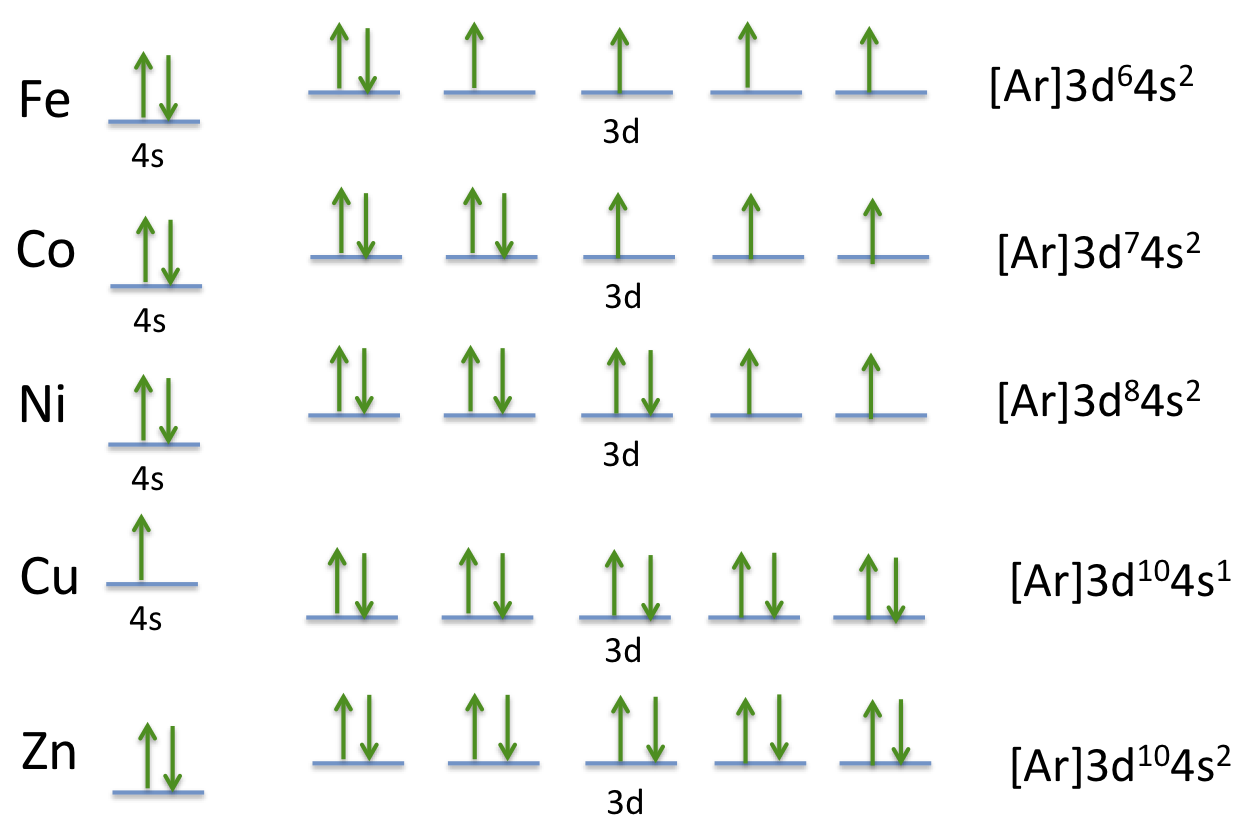
Electron Configuration Exceptions Examples: Cr, Cu, Ag, and Mo. 3+ Orbital energy diagram of Cr with no attached ligands. All five 3d-orbitals have the same energy. 3. Using your answer from Question 2, draw a d-orbital splitting diagram for low-spin Co3+ similar to the one given for Cr3+ in the introduction to this experiment. High Cr3+ doping levels, up to 8 mole percent, and low losses have been obtained with the tunable solid- state laser material LiCaAlF6:Cr3+ (Cr:LiCAF). Measurements and calculations show that high pumping and extraction efficiencies are possible with the improved material. © 1990 Optical Society of... On the other hand, further rationalizations of the kinetic rates for the other 2+ and 3+ first row transition metals requires a correlation between exchange rates Considering an octahedral geometry where the ligands act as point charges located along the cartesian axes around Cr III , the half-filled t 2g orbitals...
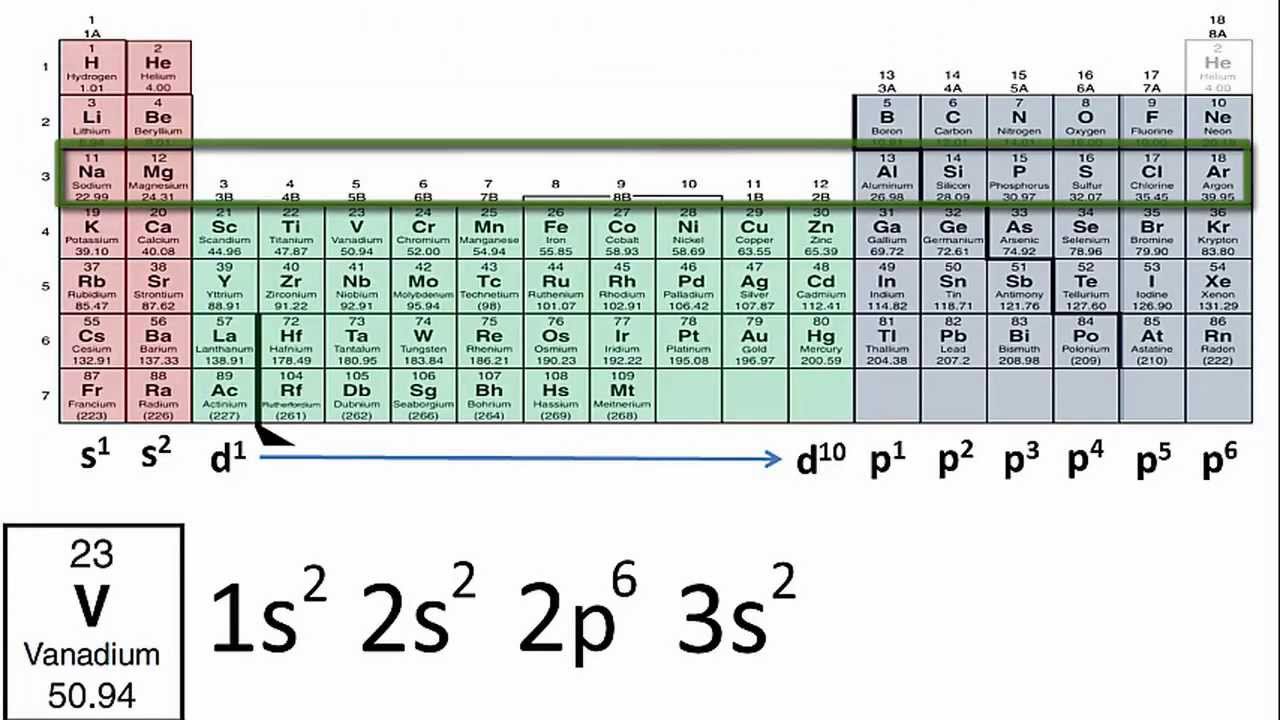
Orbital Diagrams Chemistry Tutorial. Key Concepts. An orbital diagram, or orbital box diagram, is a way of representing the electron configuration of an atom. A box, line, or circle, is drawn to represent each orbital in the electron configuration (using the Aufau Principle to order the orbitals and hence... Question: What Is The Ground State Orbital Diagram Of Cr3+? Transcribed Image Text from this Question. What is the ground state orbital diagram of Cr3+? Molecular Orbital Diagram Wikiwand Here's what i have for the electron configuration for chromium with the orbital notation. 1080x684 - It explains What Is The Electron Configuration Of Cr2 Clutch Prep Cr, cr2+, and cr3+ electron configuration notation. 798x411 - To be able to use crystal field... The Pourbaix diagram for chromium in pure water, perchloric acid, or sodium hydroxide[22][23]. Chromium is a member of group 6, of the transition Chromium(III) hydroxide (Cr(OH)3) is amphoteric, dissolving in acidic solutions to form [Cr(H2O)6]3+, and in basic solutions to form [Cr(OH) 6]3− . It is...

Solved What Is The Maximum Number Of Electrons That Can Occupy Each Of The Following A 4d 10 E 4f 1l E 6s B 5f I4 D 2p 6 F 3f Px Pyrz
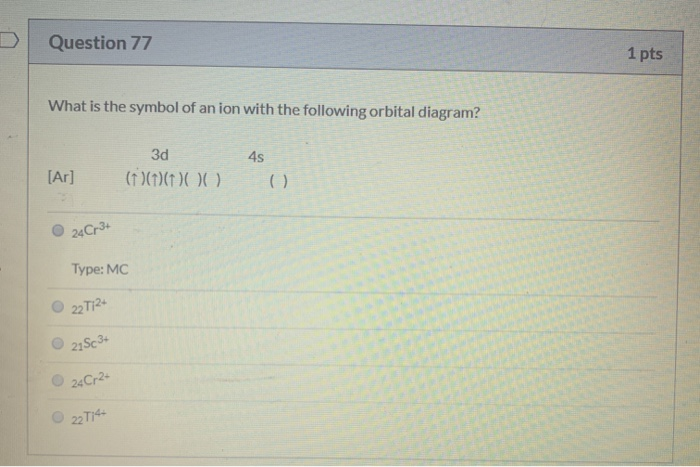
Answer to Write orbital diagrams for each of these ions. V5+,Cr3+,Ni2+,Fe3+ Determine if the ion is diamagnetic or paramagnetic. V 86%(14). V5+ orbital diagram keyword after analyzing the system lists the list of keywords related and the list of websites with related content, in addition you can see...
a. V5+ b. Cr3+ c. Ni2+ d. Fe3+. Do some orbital configurations or diagrams for some metal cat ions. So first example is going to be for chromium three plus. So as we follow the periodic table and fill the electrons properly, first row of the periodic table.
Only RUB 193.34/month. Electron Configurations and Orbital Diagrams. Terms in this set (18). C (carbon). What element does the following orbital diagram represent? Mg (magnesium).
d-Orbital Splittings. CFT focuses on the interaction of the five (n − 1)d orbitals with ligands arranged in a regular array around a transition-metal ion. We will focus on the application of CFT to octahedral complexes, which are by far the most common and the easiest to visualize.
(b) Cr3+ The Cr3+ ion is a d3 case. Both weak and strong field complexes have three unpaired. electrons. Note: you do not need to show both diagrams, as they are the same. LECTURE 28. (c) Cd2+ The Cd+2 ion is a d10 case.
Note that it is #4s^(1)3d^(5)# and not #4s^(2)3d^(4)# because a half filled #d# orbital is more stable than a partially filled #d# orbital.
› cr3+ orbital diagram. Electron Configuration for Cr, Cr2+, and Cr3+ (Exception ... See more all of the best faqs on www.pinterest.com ▼.
Partial Orbital Diagrams and Condensed Configurations. A partial orbital diagram shows only the highest energy sublevels being Write the condensed electron configuration for each atom, recalling the irregularity for Cr. Since there are 3 d electrons they are all unpaired. Cr3+ is paramagnetic.
Video: Cr, Cr2+, and Cr3+ Electron Configuration Notation. • In writing the electron configuration for Chromium the first two electrons will go in the 1s orbital.
Chromium(III) ion. Molecular Formula Cr. Average mass 51.994 Da. Cr3+. Chromic cation. Chromium ion.

Show The Orbital Diagrams For The Following Chromium Atom And Chromium Iii Ion Include Only Occupied Orbital S A Cr B Cr 3 Study Com

1 Write Orbital Diagrams For Each Of These Ions A V5 B Cr3 C Ni2 D Fe3 2 Determine If The Ion Is Diamagnetic Or Paramagnetic A V5 B Cr3 C Ni2







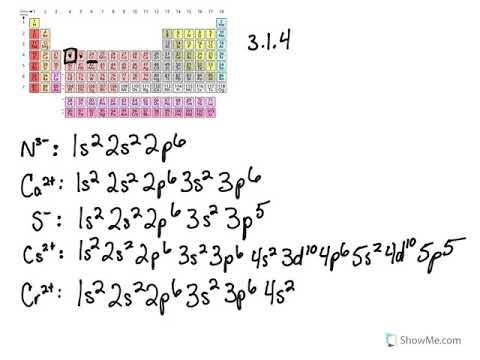











0 Response to "37 cr3+ orbital diagram"
Post a Comment